Research Highlights
Get3/4/5 Complex
Yeast chaperone protein complex
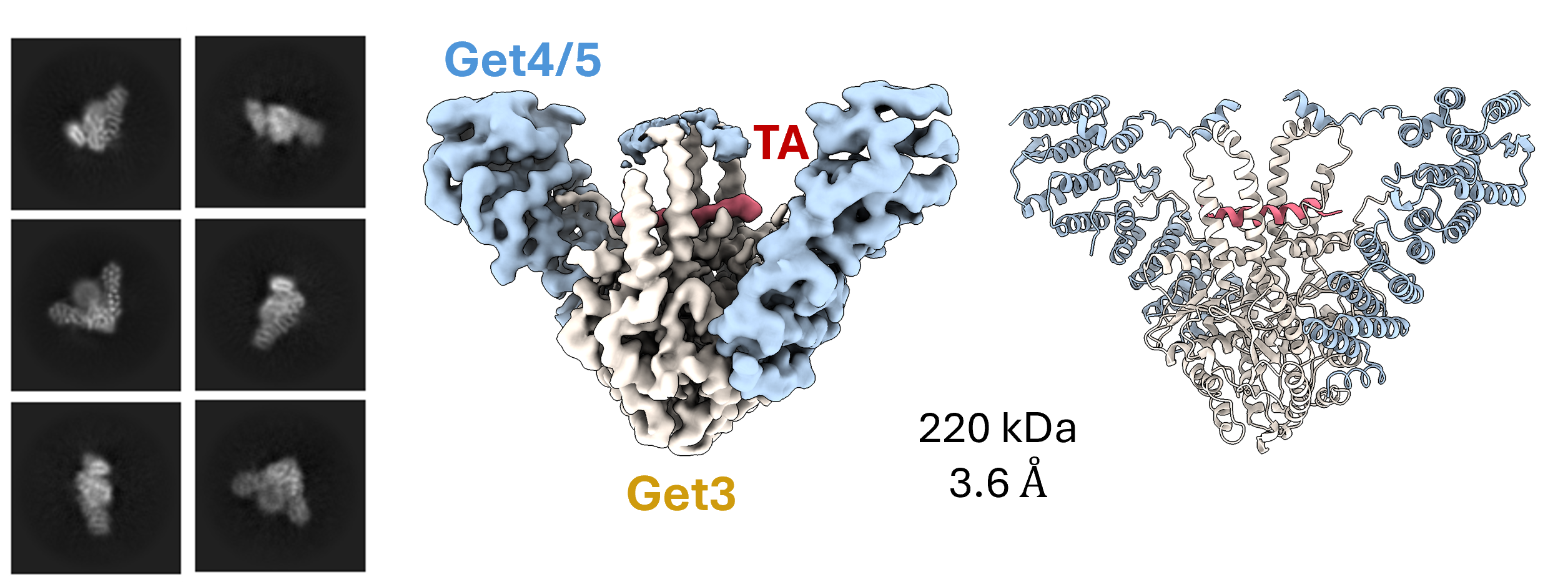
Data from the Kim lab collected at the MSU Cryo-EM RTSF Core Facility. Cells depend on precise delivery systems to ensure newly made membrane proteins reach the right destination. One factor responsible for this task is Get3, which delivers a class of tail-anchored membrane proteins (TAs) to the endoplasmic reticulum (ER). Cryo-EM structures of the Get3/4/5 determined in the absence and presence of the TA substrate revealed the underlying molecular mechanism of how Get3 relies on another protein complex, Get4/5, to capture TAs and prepare for loading. Read more here.
AaaA
Autotransporter in P. aeruginosa
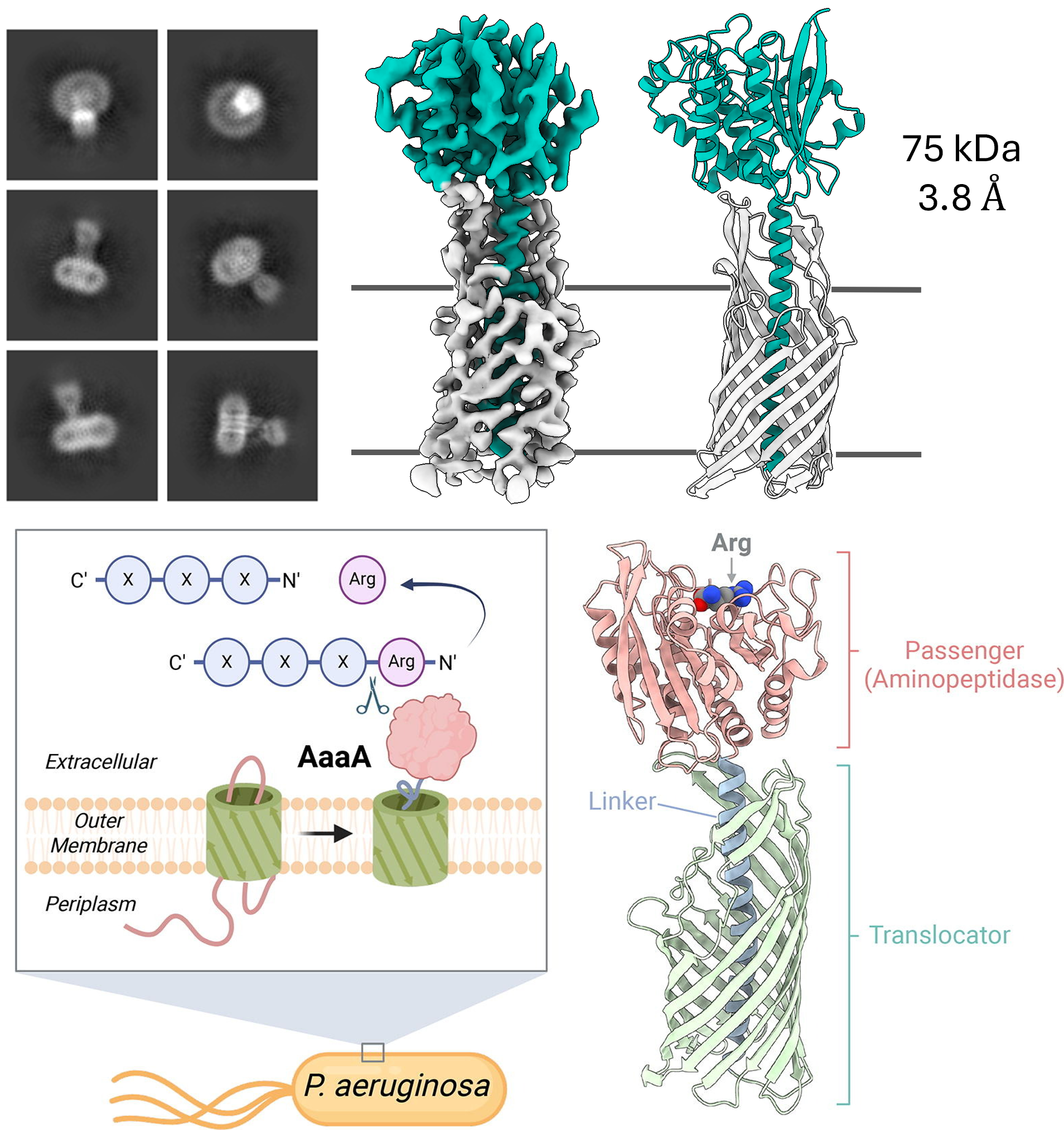
Data from the Kim lab collected at the MSU Cryo-EM RTSF Core Facility. AaaA is a virulence-associated outer membrane protein found in the Gram-negative pathogen Pseudomonas aeruginosa. Classified as both an autotransporter and a member of the M28 family of aminopeptidases, AaaA has been shown to cleave N-terminal arginine residues from host-derived peptides. This activity has been demonstrated to enhance bacterial survival and suppress host immune responses by increasing local arginine availability. Read more here.
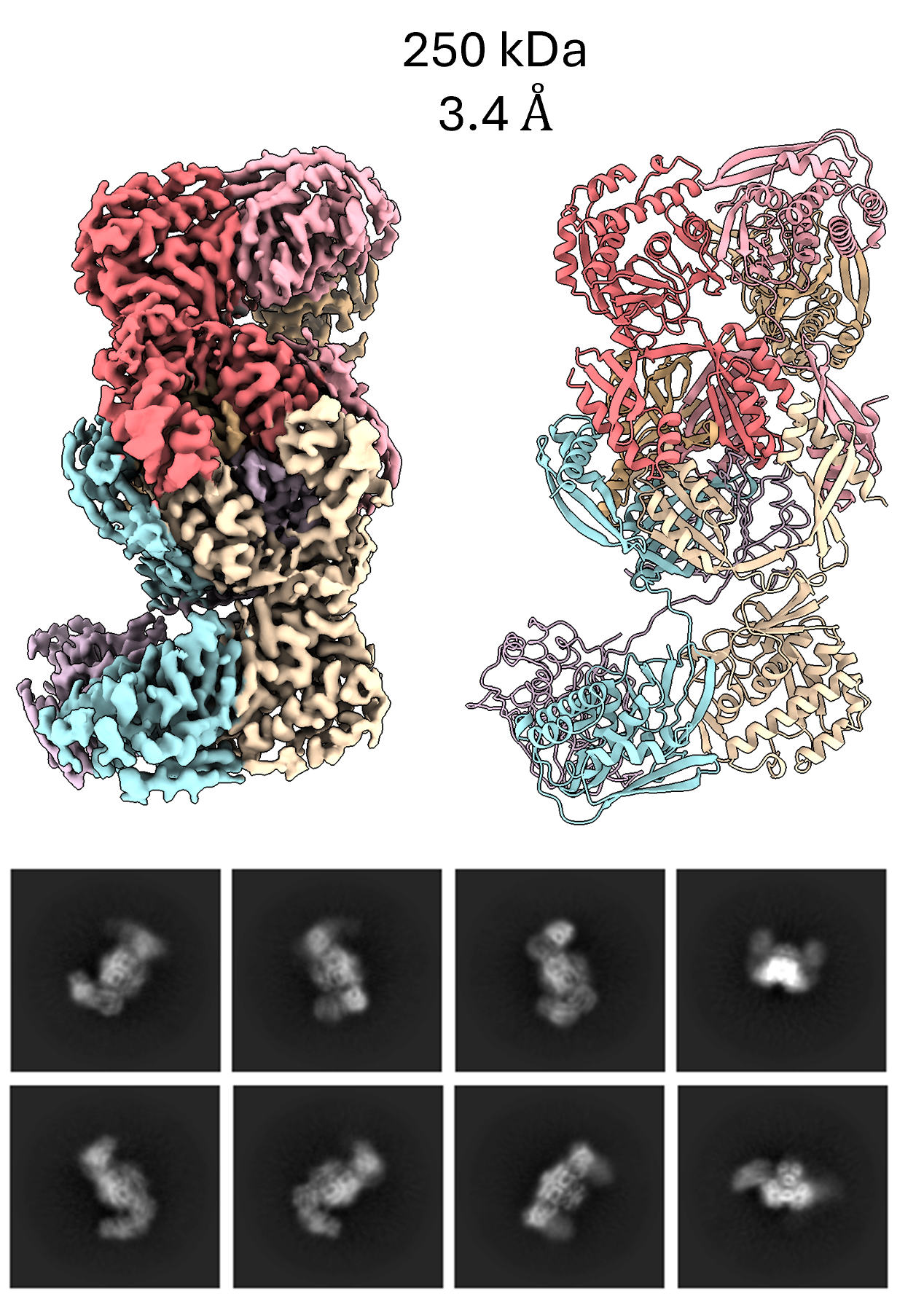
Data from the Kim lab collected at the MSU Cryo-EM RTSF Core Facility. LarC is the only known cyclometallase in nature that catalyzes the final step of the nickel-pincer nucleotide (NPN), is a recently discovered biological metallocofactor whose biosynthesis uniquely requires the formation of a stable covalent nickel-carbon bond. The first full-length LarC structure (from Moorella thermoacetica) determined by cryo-EM has provided important insights into understanding how substrate binding, nickel coordination, and CTP utilization are coordinated during catalysis.
Guinea Pig Mitochondria with Large Calcium Granules
Tomogram and Segmentation model
Data from the Bazil lab collected at the MSU Cryo-EM RTSF Core Facility.